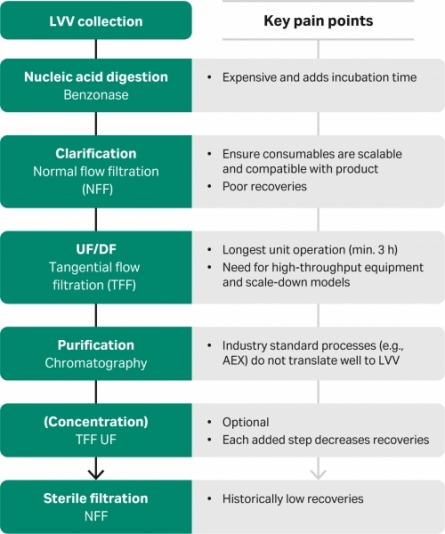
Lentiviral vectors (LVV) are a common vehicle to deliver genetic material in chimeric antigen receptor (CAR) T-cell therapy and gene therapy applications. Production methods have been developed by adapting technologies from the bioprocessing sector. However, these downstream workflows are long, require substantial manual labor, and suffer from low yields of infectious virus. This article discusses process development pain points of a modern workflow and ways to address them.