Continuous unit operations have been used successfully in manufacturing of several approved products with a collective revenue of $25 billion annually. Many large biopharmaceutical organizations are exploring potential solutions to drive continuous operations on the manufacturing floor.
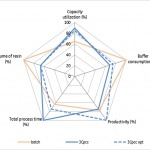
Continuous biomanufacturing carries the hope for achieving the next level of efficiency through significant increases in productivity, floor space reduction, and lower capital cost for manufacturing facilities as well as lower overall production costs.
Adopting advanced process design will need an accurate business case and risk profile for implementation. Within the risk profile, assurance of patient safety and supply through product quality demonstration will be critical.
This poster will highlight trade-offs and benefits associated with the adoptions of connected and continuous manufacturing.